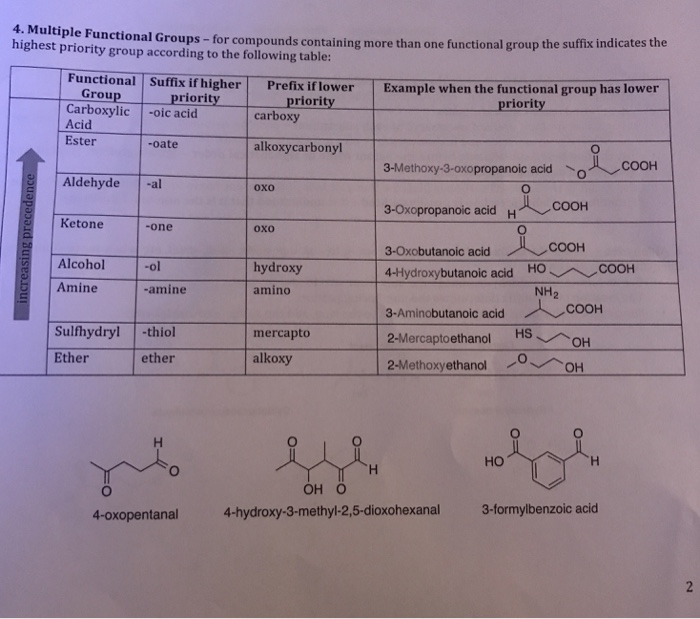
When you add any halogen “-X” (Group 7a: -F, -Cl, -Br, -I) you get an alkyl halide (RX).List of Functional Groups Without Carbonyls You’ll see soon why the “no carbonyls” part is important in a sec. When you add single bonded heteroatoms (non-carbon atoms) to alkane chains, you end up getting a bunch of the most essential functional groups for organic chemistry. Some textbooks call them types of “moieties” (a bonding pattern) but I’m just going to call them carbonyls and expect you to remember that they are not functional groups.Ĭarbonyl is not Technically a Functional Group They are NOT true functional groups since they are an essential component of many different functional groups. Intermission: What is a carbonyl and why does it matter?Ĭarbonyls are just C=O bonds on your molecule. Now that we are done with hydrocarbons, let’s talk about carbonyls. These names are counterintuitive, since it makes logical sense that benzene connected to a main chain is called “benzyl”, but chemists aren’t always logical and that’s just plain wrong. When you extend benzene off of a carbon chain (-R), you get the side chain functional groups phenyl (directly attached to the main chain) and benzyl (extra –CH2 between the benzene and the main chain). Remember: The “-R” symbol is used to represent extra hydrocarbon side chains on the molecule that we don’t particularly care about at that moment. We will learn more about the names of benzene derivatives later in the year. Thus, benzene is part of an aromatic functional group. The group is actually called aromatics or arenes. This is how we get alkenes (C=C double bonds) and alkynes (C≡C triple bonds).īenzene is the name of a 6-membered ring with alternating single and double bonds (like the Clutch Prep Logo), however “benzene” is not really a functional group since it is the name of a single molecule. However, by mixing carbons and hydrogens together, you can get a few other combinations of carbon and hydrogen that are uniquely reactive. For that reason, alkanes are not actually considered functional groups, since they provide the backbone for everything else. Hydrocarbons: Starting with the BackboneĪlkanes (C-C single covalent bonds) are the skeleton of organic chemistry. PART 1: MEMORIZING THE 17 MOST IMPORTANT FUNCTIONAL GROUPS 1. 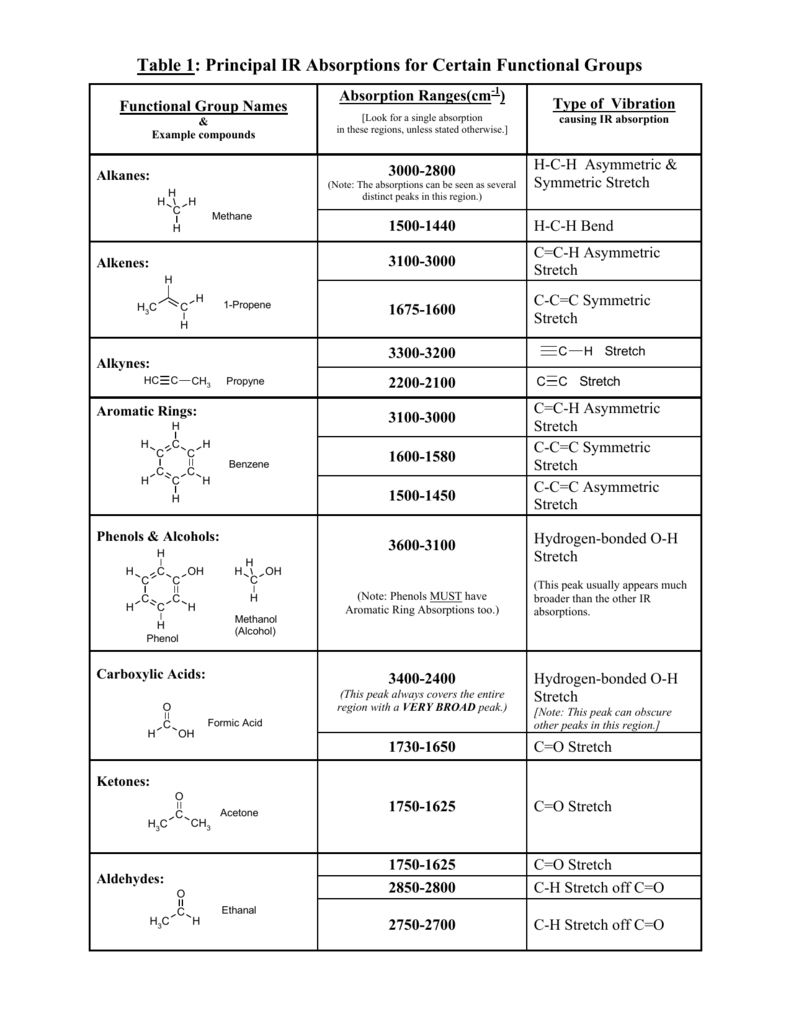
If you still need help with understanding these, feel free to watch my videos on skeletal structure. Meaning I’ll be dropping all hydrogens and lone pairs. HEADS UP: For this topic, I’m going to assume you already know how to represent molecules in skeletal (bond line/line angle) form. (In Orgo 2 the list gets longer, and I’ll have a separate post for those.) I’m about to give you the functional group run-down of your life, with examples and charts of how to memorize these. Thankfully, in undergraduate organic chemistry 1 you’re only going to be responsible to know ~17 of these specific groups. Specifically, when compared against each other, they share similarities in polarity, acidity, chemical reactivity, etc. We could spend the next 100 years learning the name and reactivity of each molecule separately OR we could try to find some patter ns in the chaos. Why do we even need functional groups? There are quite literally infinite combinations of molecules found in the universe. 
The 17 most Important Functional Groups Chart In this post I’m going to teach you how to memorize them based on this chart: You’ll need many of these later, for example when you study carbohydrates and protein functional groups in biology. INTRO: WHY DO WE NEED FUNCTIONAL GROUPS?įunctional groups are some of the first terms you learn to recognize in undergraduate organic chemistry- and they happen to be some of the most important. P.S.S Here is the 49-minute super-comprehensive version if you are not in a time crunch. Here’s my 5-minute video on memorizing the 17 most important functional groups. By definition, functional groups are specific groups of atoms commonly found across molecules all over the universe, that look and behave similarly when exposed to like conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
